The adiabatic process is a fundamental concept in thermodynamics, describing a specific type of thermodynamic transformation in which a system exchanges no heat with its surroundings. This process is crucial for understanding various physical phenomena, including the behavior of gases, the operation of heat engines, and the principles of refrigeration. This article will provide an in-depth examination of the adiabatic process, covering its definition, characteristics, equations, types, applications, and illustrative explanations to enhance understanding.
1. Definition of Adiabatic Process
An adiabatic process is defined as a thermodynamic process in which there is no heat transfer (![]() ) between a system and its surroundings. This means that any change in the internal energy of the system is solely due to work done on or by the system. The term “adiabatic” comes from the Greek word “adiabatos,” meaning “impassable,” indicating that heat cannot pass into or out of the system during the process.
) between a system and its surroundings. This means that any change in the internal energy of the system is solely due to work done on or by the system. The term “adiabatic” comes from the Greek word “adiabatos,” meaning “impassable,” indicating that heat cannot pass into or out of the system during the process.
Illustrative Explanation: Imagine a perfectly insulated thermos bottle filled with hot coffee. If you seal the thermos and leave it for a while, the coffee will remain hot because no heat can escape (adiabatic condition). If you were to open the thermos, heat would flow out, and the coffee would cool down, which would no longer be an adiabatic process.
2. Characteristics of Adiabatic Processes
Adiabatic processes have several key characteristics that distinguish them from other thermodynamic processes:
- No Heat Transfer: As mentioned, the defining feature of an adiabatic process is that there is no heat exchange with the surroundings. This is often achieved through rapid processes or perfect insulation.
- Change in Internal Energy: In an adiabatic process, the change in internal energy (
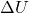 ) of the system is equal to the work done on or by the system. This relationship can be expressed as:
) of the system is equal to the work done on or by the system. This relationship can be expressed as:
![]()
where ![]() is the work done.
is the work done.
- Temperature Change: Since there is no heat transfer, the temperature of the system can change as work is done. For example, compressing a gas adiabatically will increase its temperature, while expanding it will decrease its temperature.
Illustrative Explanation: Consider a bicycle pump. When you compress the air inside the pump by pushing the handle down quickly, the air heats up (temperature change) because no heat is exchanged with the environment (adiabatic). If you were to compress the air slowly, heat could escape, and the temperature might not rise as much, indicating a non-adiabatic process.
3. Equations Governing Adiabatic Processes
The behavior of an ideal gas undergoing an adiabatic process can be described using specific equations derived from the first law of thermodynamics and the ideal gas law. For an ideal gas, the following relationships hold:
- Adiabatic Condition: The relationship between pressure (
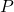 ), volume (
), volume (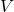 ), and temperature (
), and temperature (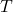 ) for an ideal gas undergoing an adiabatic process is given by:
) for an ideal gas undergoing an adiabatic process is given by:
![]()
where ![]() (gamma) is the heat capacity ratio, defined as:
(gamma) is the heat capacity ratio, defined as:
![]()
Here, ![]() is the heat capacity at constant pressure, and
is the heat capacity at constant pressure, and ![]() is the heat capacity at constant volume.
is the heat capacity at constant volume.
- Temperature-Volume Relationship: The relationship between temperature and volume during an adiabatic process is given by:
![]()
- Pressure-Temperature Relationship: The relationship between pressure and temperature during an adiabatic process is given by:
![]()
Illustrative Explanation: Imagine a balloon filled with air. If you squeeze the balloon quickly (adiabatic compression), the pressure inside increases, and the temperature rises. The equations above describe how the pressure, volume, and temperature of the gas inside the balloon change during this process, similar to how the balloon behaves when you apply force.
4. Types of Adiabatic Processes
Adiabatic processes can be classified into two main types based on the nature of the work done:
- Adiabatic Expansion: This occurs when a gas expands without exchanging heat with its surroundings. During adiabatic expansion, the gas does work on its surroundings, leading to a decrease in internal energy and temperature.
- Adiabatic Compression: This occurs when a gas is compressed without heat exchange. In this case, work is done on the gas, resulting in an increase in internal energy and temperature.
Illustrative Explanation: Think of a piston in a cylinder filled with gas. If you pull the piston out quickly (adiabatic expansion), the gas expands, does work on the piston, and cools down. Conversely, if you push the piston in quickly (adiabatic compression), the gas is compressed, work is done on it, and it heats up.
5. Applications of Adiabatic Processes
Adiabatic processes have numerous applications across various fields, including:
- Heat Engines: Many heat engines, such as internal combustion engines and gas turbines, operate on cycles that include adiabatic processes. Understanding these processes is essential for optimizing engine performance and efficiency.
- Refrigeration and Air Conditioning: Adiabatic processes are utilized in refrigeration cycles, where gases are compressed and expanded to absorb and release heat, effectively cooling the environment.
- Meteorology: In meteorology, adiabatic processes are crucial for understanding atmospheric phenomena. For example, when air rises in the atmosphere, it expands adiabatically, leading to cooling and cloud formation.
- Thermodynamic Cycles: Adiabatic processes are integral to various thermodynamic cycles, such as the Carnot cycle and the Brayton cycle, which are fundamental to understanding energy conversion and efficiency.
Illustrative Explanation: Consider a car engine. When the fuel-air mixture is compressed in the cylinder (adiabatic compression), it heats up, leading to combustion. The rapid expansion of gases pushes the piston down (adiabatic expansion), converting thermal energy into mechanical work. Similarly, in a refrigerator, the refrigerant gas is compressed and expanded to absorb heat from the inside, keeping your food cool.
6. Limitations of Adiabatic Processes
While the adiabatic process is a useful idealization, it has limitations:
- Real Gases: In real-world applications, gases may not behave ideally, especially at high pressures and low temperatures. Deviations from ideal behavior can affect the accuracy of predictions based on adiabatic processes.
- Heat Transfer: In practical situations, it is challenging to achieve perfect insulation. Some heat transfer may occur, leading to deviations from the ideal adiabatic process.
- Time Factor: The speed of the process is crucial. If the process occurs too slowly, heat may be exchanged with the surroundings, making it non-adiabatic.
Illustrative Explanation: Imagine trying to keep a pot of soup hot by wrapping it in a thick blanket (attempting to create an adiabatic condition). If you leave it for too long, some heat will escape, and the soup will cool down. Similarly, in real-world applications, achieving a perfect adiabatic process is often impractical due to heat loss and the behavior of real gases.
7. Conclusion
The adiabatic process is a fundamental concept in thermodynamics that describes heat transfer without heat exchange with the surroundings. By exploring its definition, characteristics, governing equations, types, applications, and limitations, we gain a deeper appreciation for its significance in various scientific and engineering contexts. From heat engines to refrigeration systems and atmospheric phenomena, adiabatic processes play a vital role in understanding energy transfer and conversion. Whether in theoretical studies or practical applications, the principles of adiabatic processes remain essential for advancing our knowledge of thermodynamics and improving the efficiency of energy systems. Understanding adiabatic processes allows engineers, scientists, and researchers to design better systems and technologies, ultimately contributing to advancements in various fields.